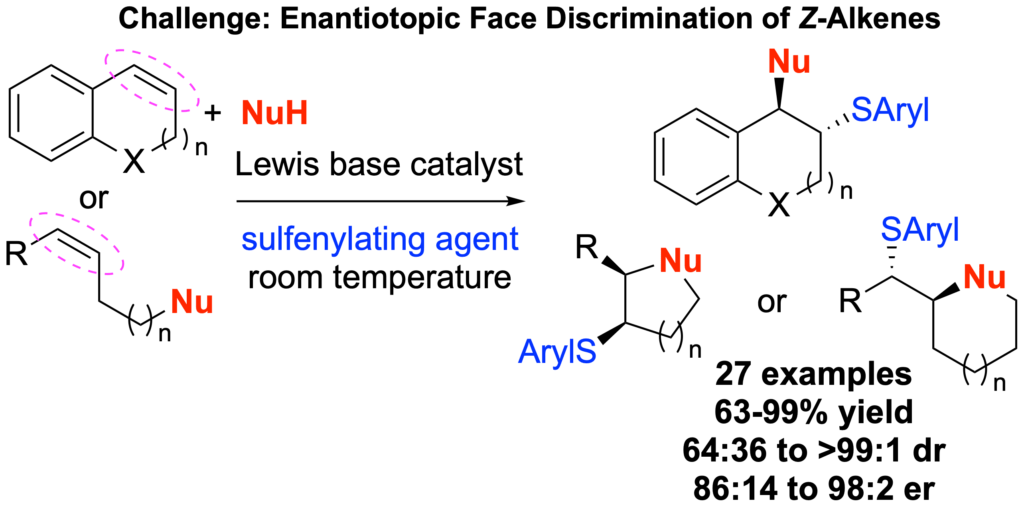
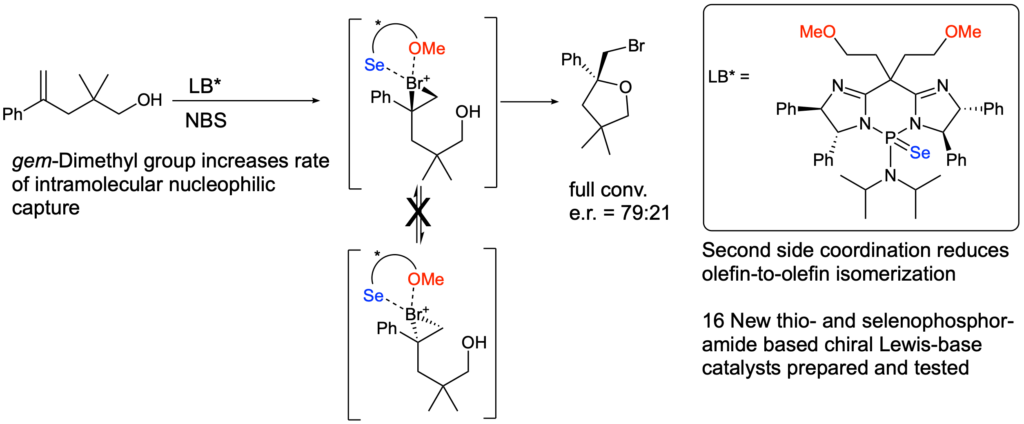
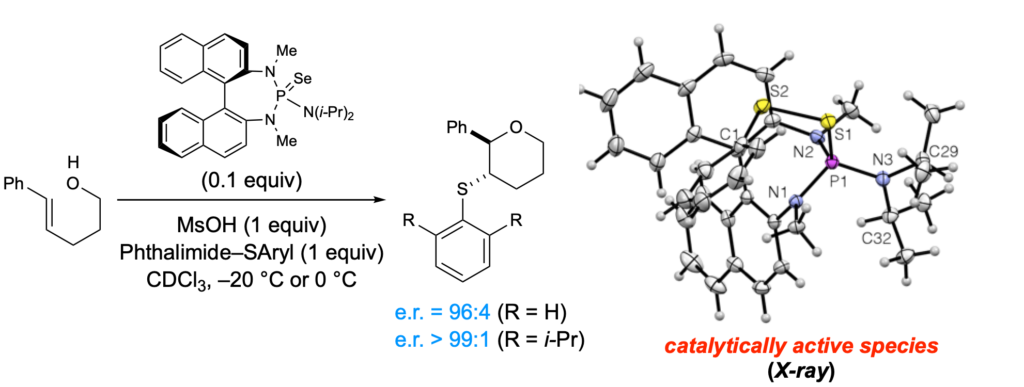
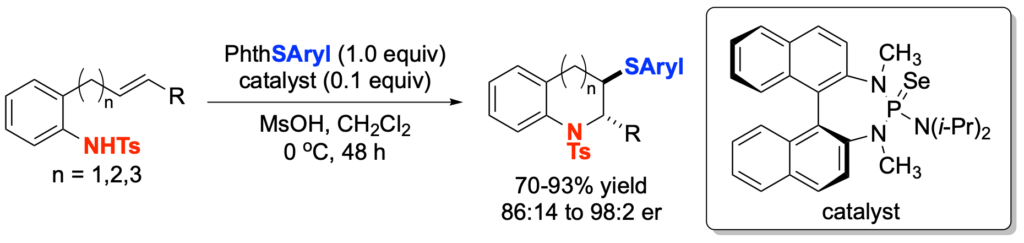
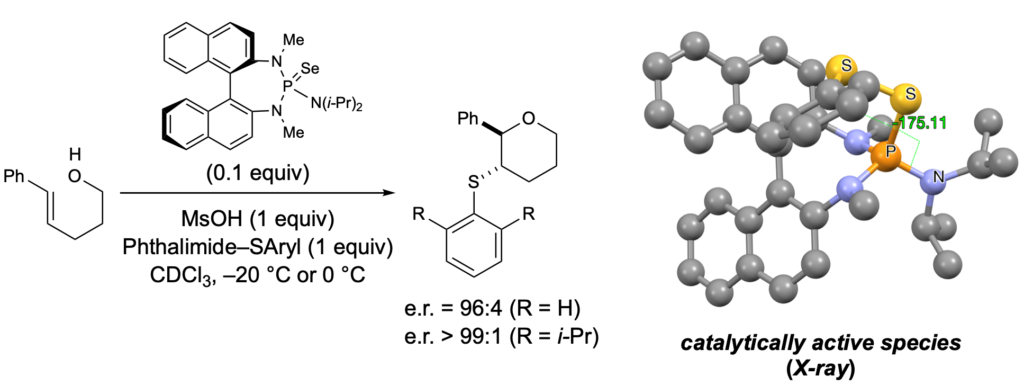
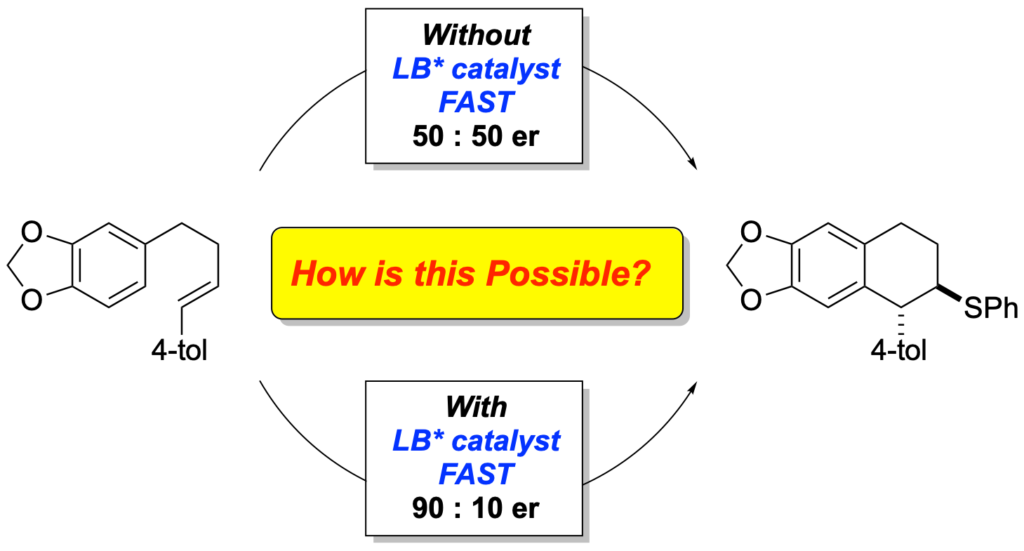
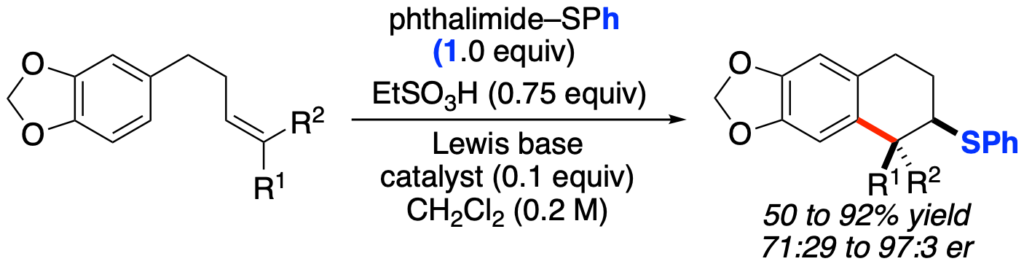
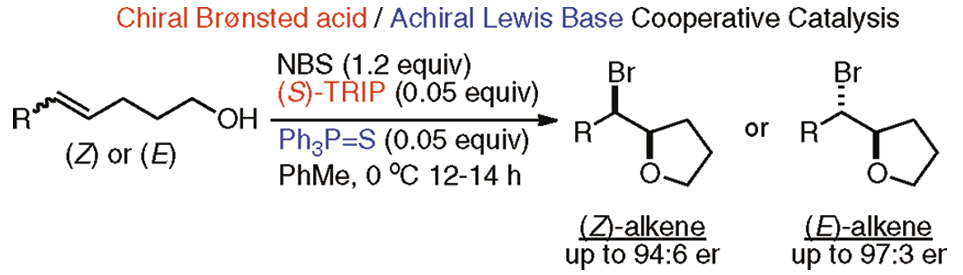
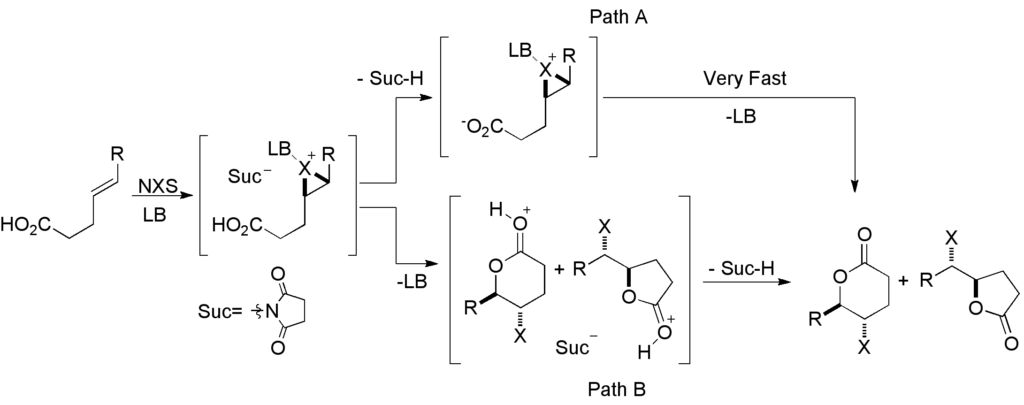
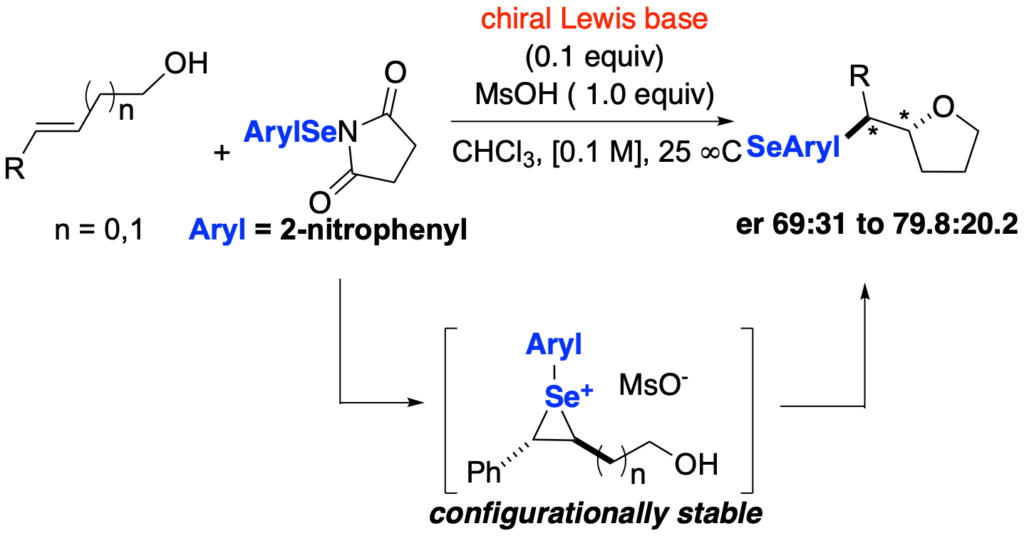
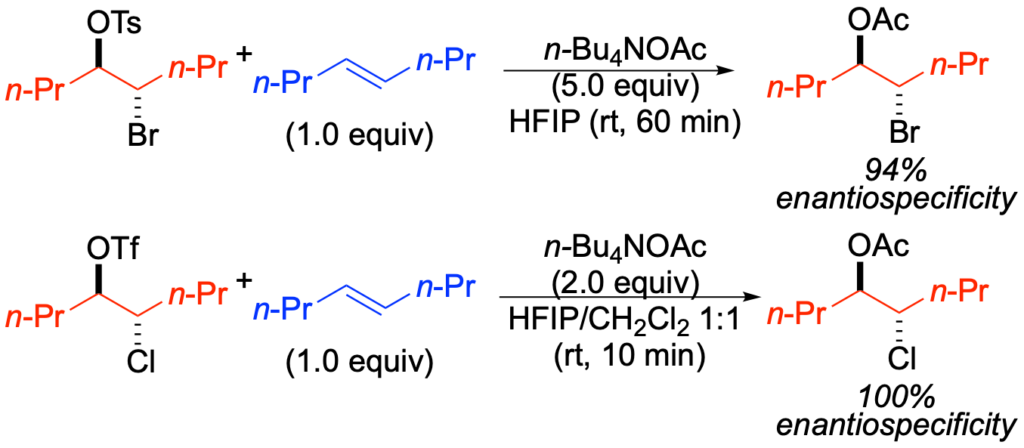
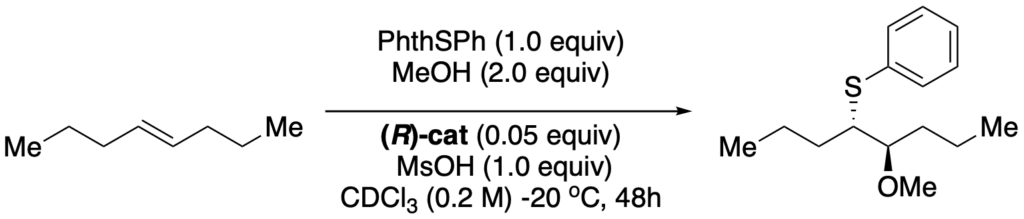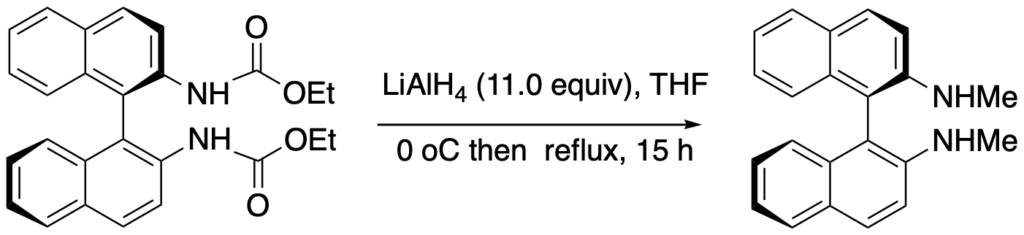[419] Enantioselective Inter- and Intramolecular Sulfenofunctionalization of Unactivated Cyclic and Z-Alkenes (with A. Matviitsuk, J. L .Panger) ACS Catalysis, 2022, 12, 7377-7385.
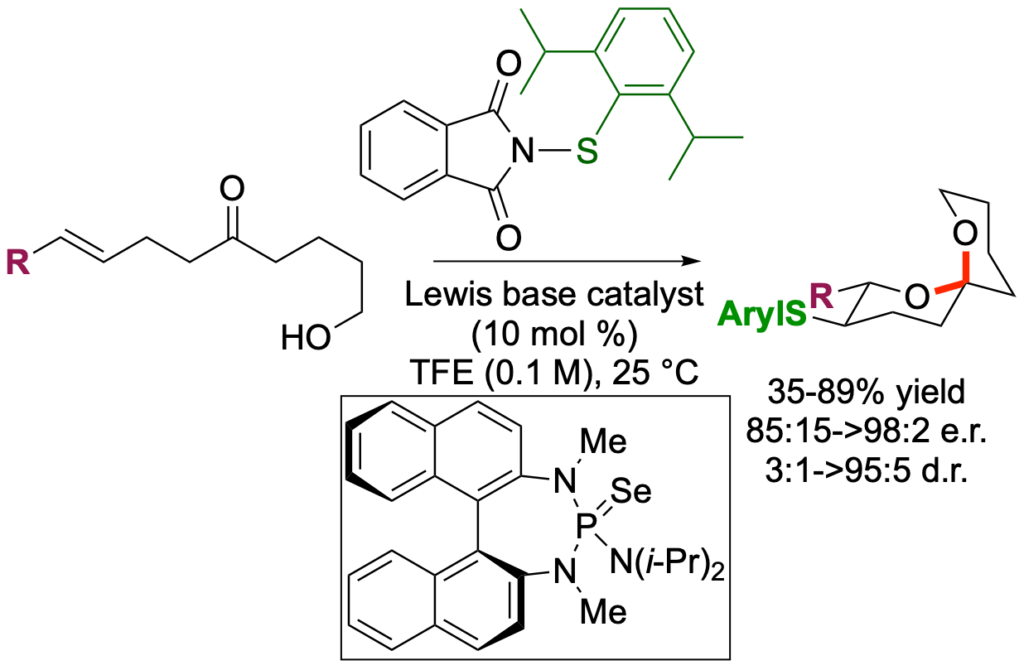
[415] Lewis Base Catalyzed, Sulfenium Ion Initiated Enantioselective, Spiroketalization Cascade (with K. M. Hilby)
J. Org. Chem. 2021, 86, 14250-14289.
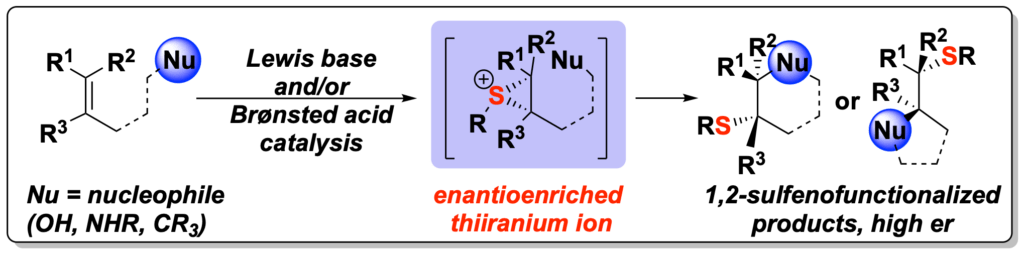
[404] Catalytic, Enantioselective Sulfenofunctionalization of Alkenes— Development and Recent Advances (with A. Matviitsuk, J. L. Panger) Angew. Chem., Int. Ed. 2020, 59, 19796 – 19819.
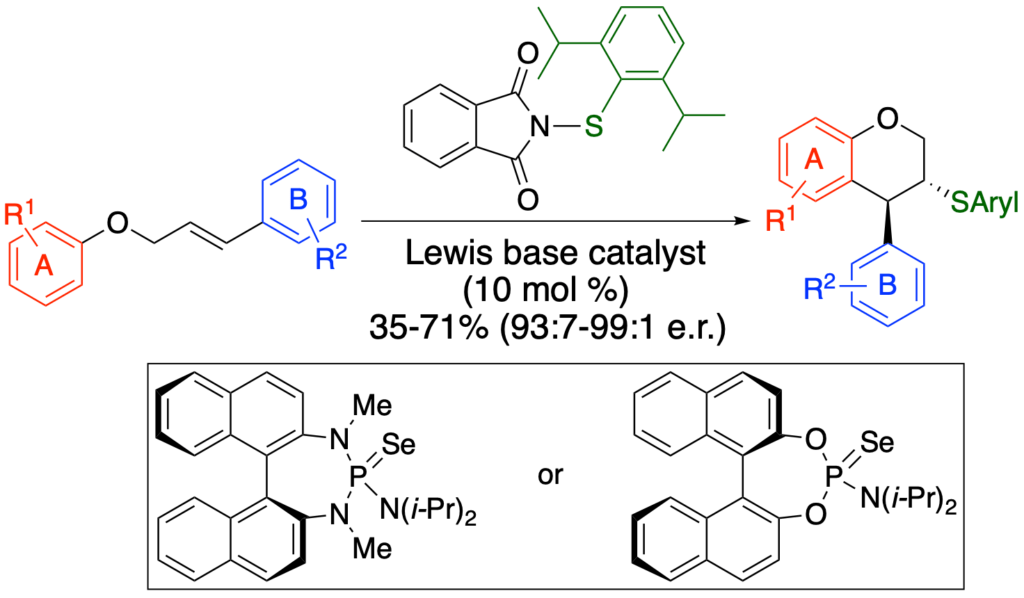
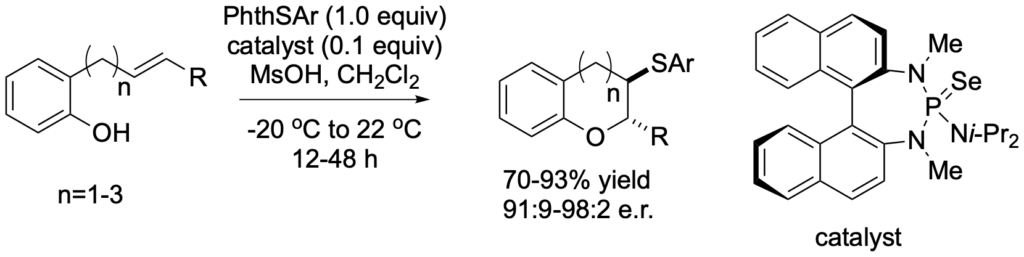
[416] Synthesis of Enantioenriched 3,4-Disubstituted Chromans through
Lewis Base Catalyzed Carbosulfenylation (with T. Menard and A. Laverny) J. Org. Chem. 2021, 86, 14290-14310.
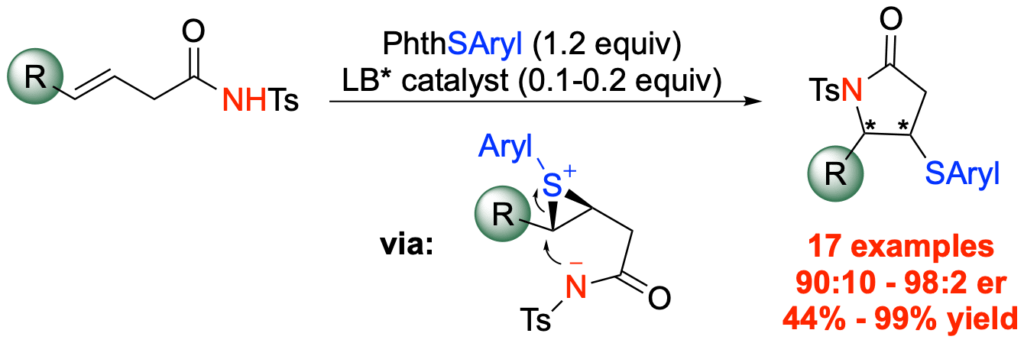
[400] Enantioselective Synthesis of γ‑Lactams by Lewis Base Catalyzed Sulfenoamidation of Alkenes (with J. L. Panger) Org. Lett. 2020, 22, 2501−2505.
[392] (R)-N,N‘-Dimethyl-1,1’-binaphthyldiamine (with P. Ryabchuk) Org. Synth. 2019, 96, 382-399.
[391] Enantioselective, Lewis Base-Catalyzed, Intermolecular Sulfenoamination of Alkenes (with A. Roth) J. Am. Chem. Soc. 2019, 141, 13767−13771.
[389] Enantio- and Diastereoselective, Lewis Base Catalyzed, Cascade Sulfenoacetalization of Alkenyl Aldehydes (with A. Matviitsuk) Angew. Chem. Int. Ed. 2019, 58, 12486 –12490.
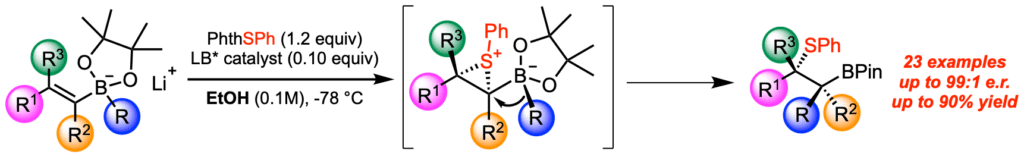
[384] Enantioselective, Lewis Base-Catalyzed Carbosulfenylation of Alkenylboronates by 1,2- Boronate Migration (with Z. Tao, K. A. Robb, J. L. Panger) J. Am. Chem. Soc. 2018, 140, 15621-15625.
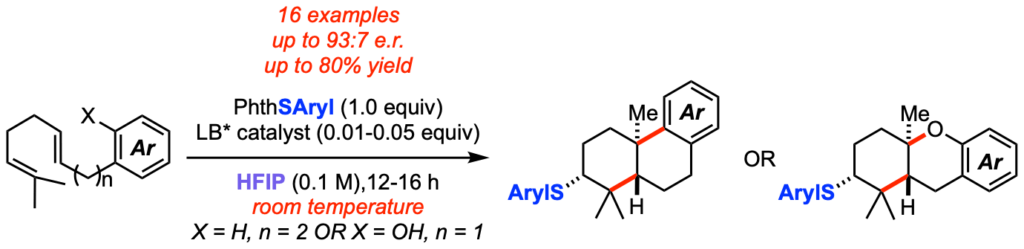
[379] Enantioselective, Lewis Base-Catalyzed Sulfenocyclization of Polyenes (with Z. Tao, K. A. Robb, K. Zhou) J. Am. Chem. Soc. 2018, 140, 3569-3573.
[373] Structural, Mechanistic, Spectroscopic, and Preparative Studies on the Lewis Base Catalyzed, Enantioselective Sulfenofunctionalization of Alkenes (with E. Hartmann) Helv. Chim. Acta 2017, 100, e1700158.
[371] Catalytic, Enantioselective, Intramolecular Sulfenoamination of Alkenes with Anilines (with H. M. Chi) J. Org. Chem. 2017, 82, 3826-3843.
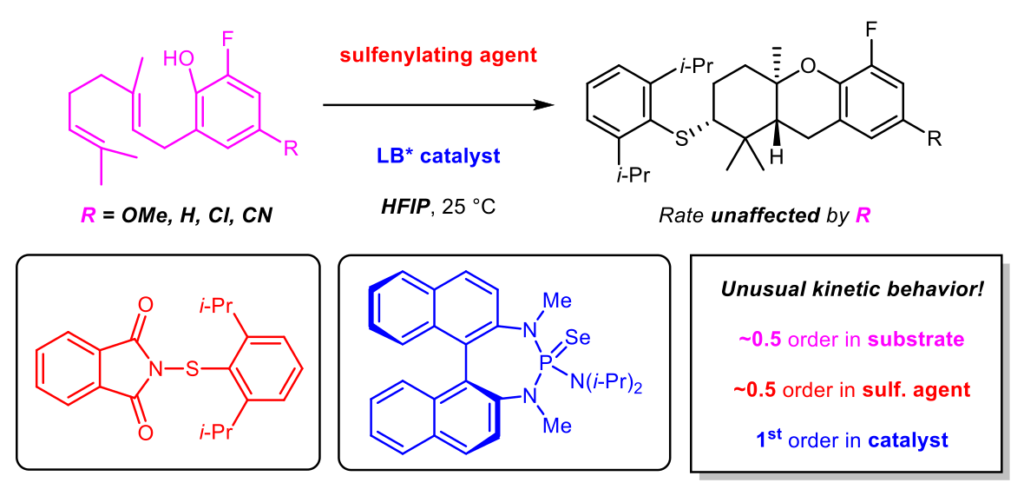
[370] Catalytic, Enantioselective, Intramolecular Sulfenofunctionalization of Alkenes with Phenols (with D. J. P. Kornfilt) J. Org. Chem. 2017, 82, 3192-3222.
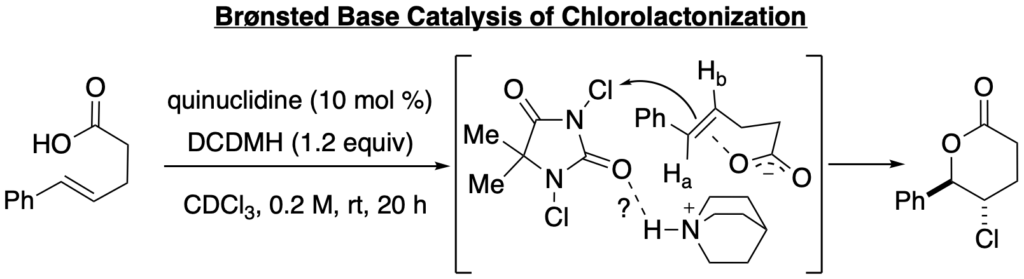
[367] Toward Catalytic, Enantioselective Chlorolactonization of 1,2-Disubstituted Styrenyl Carboxylic Acids (with P. Ryabchuk, Matthew T. Burk, and B. B. Gilbert) J. Org. Chem. 2016, 81, 10411-10423.
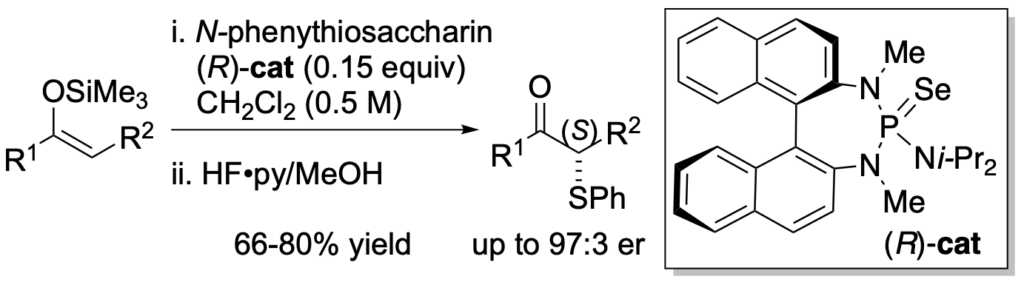
[354] Catalytic, Enantioselective Sulfenylation of Ketone-Derived Enoxysilanes (with S. Rossi, M. P. Webster, and H. Wang) J. Am. Chem. Soc. 2014, 136, 13016-13028.
[349] Lewis Base Catalyzed, Enantioselective, Intramolecular Sulfenoamination of Olefins (with H. M. Chi) J. Am. Chem. Soc. 2014, 136, 8915-8918.
[348] Catalytic, Enantioselective, Intramolecular Carbosulfenylation of Olefins. Mechanistic Aspects: A Remarkable Case of Negative Catalysis (with H. M. Chi) J. Am. Chem. Soc. 2014, 136, 3655-3663.
[346] Catalytic, Enantioselective, Intramolecular Carbosulfenylation of Olefins. Preparative and Stereochemical Aspects (with A. Jaunet) J. Org. Chem. 2014, 79, 140-171.
[341] Catalytic, Enantioselective, Intramolecular Carbosulfenylation of Olefins (with A. Jaunet) J. Am. Chem. Soc. 2013, 135, 6419-6422.
[333] Enantioselective Bromocycloetherification by Lewis Base/Chiral Brønsted Acid Cooperative Catalysis (with M. T. Burk) Org. Lett., 2012, 14, 256-259.
[331] Catalytic Asymmetric Thiofunctionalization of Unactivated Alkenes (with D. J. P. Kornfilt and T. Vogler) J. Am. Chem. Soc. 2011, 133, 15308-15311.
[322] Lewis Base Catalysis of Bromo- and Iodolactonization, and Cycloetherification (with M. T. Burk) Proc. Nat. Acad. Sci. 2010, 107, 20655-20660.
[320] Preparative and Mechanistic Studies toward the Rational Development of Catalytic, Enantioselective Selenoetherification Reactions (with D. Kalyani and W. R. Collins) J. Am. Chem. Soc. 2010, 132, 15752-15765.
[311] On the Absolute Configurational Stability of Bromonium and Chloronium Ions (with M. T. Burk and A. J. Hoover) J. Am. Chem. Soc. 2010, 132, 1232-1233.
[309] Synthesis and Reactivity of Enantiomerically Enriched Thiiranium Ions (with T. Vogler) Chem. Eur. J. 2009, 15, 11737-11745.
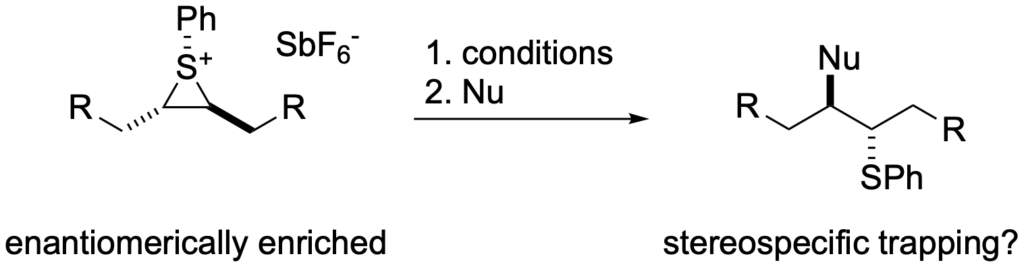
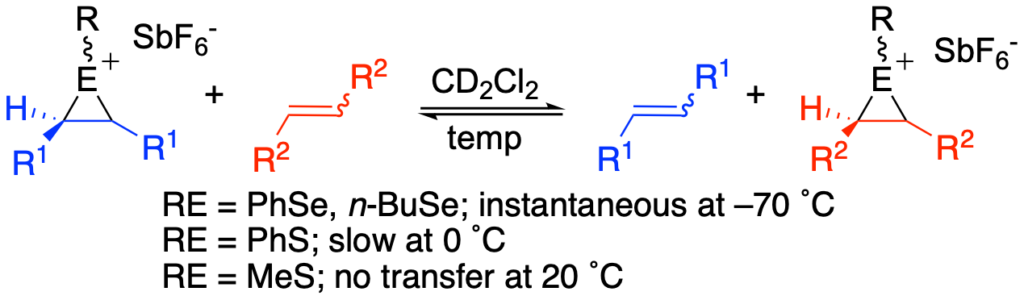
[302] Observation of Direct Sulfenium and Selenenium Group Transfer from Thiiranium and Seleniranium Ions to Alkenes (with W. R. Collins and M. D. Cullen) J. Am. Chem. Soc. 2009, 131, 3490-3492.
[277 ] Lewis Base Activation of Lewis Acids: Development of a Lewis Base Catalyzed Selenolactonization (with W. R. Collins) Org. Lett. 2007, 9, 3801-3804.
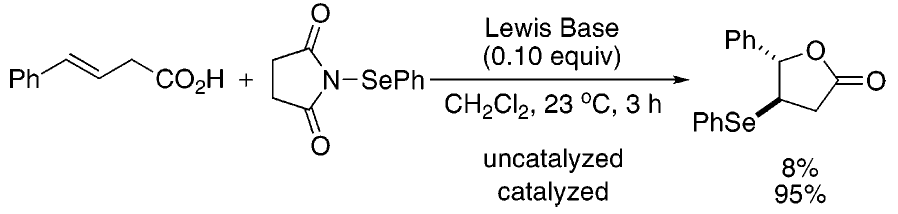
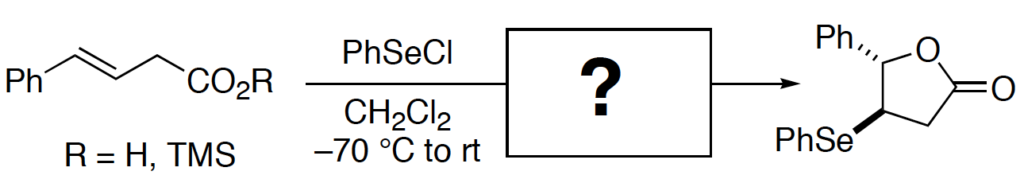
[265] On the Mechanism of the Selenolactonization Reaction with Selenenyl Halides (with M. G. Edwards) J. Org. Chem. 2006, 71, 7293-7306.
- Group 16 and 17 Lewis Base Catalysis
- Syn-Difunctionalization of Alkenes
- Chemoinformatics
- Boron-Based Cross-Coupling Reactions
- Silicon-Based Cross-Coupling Reactions
- Water-Gas Shift Reaction
- Phase Transfer Catalysis
- Organoelement Chemistry
- Allylmetal Aldehyde and Acetal Reactions
- Aldol Chemistry
- Nitroalkene and Related Cycloaddition (Including Tandem)
- Chemistry of Phosphorus-Stabilized Anions
- Dioxirane Chemistry
- Silicon-Directed Nazarov Cyclization and Related Reactions
- Carbanion-Accelerated Claisen Rearrangements
- Perspectives and Reviews
- Miscellaneous